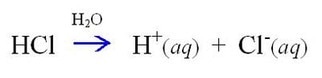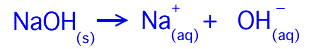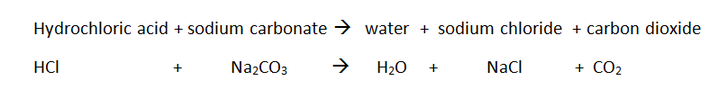ACIDS AND BASES
1. Where do we find acids and bases?
The short answer is everywhere! Life itself is likely to have originated from the movement of acidic ions in an out of a primitive cell.
Every liquid you see will probably have either acidic or basic properties. One exception might be distilled water. Distilled water is just water. The acidic and basic ions in distilled water are in equal amounts and cancel each other out. Most water you drink has an excess of acidic or basic ions in it.
Acids may occur naturally while some are synthetic. They are all characterised by their distinctive sour taste, such as in lemon juice or vinegar and some insect stings, although in many cases it is not recommended to taste them. Not all acids are dangerous but they can be corrosive and dissolve ('eat-away') a number of solid material such as metal, glass, rocks, clothing or burn skin.
The photo below shows some of the many natural acids found in food and drink. For example, The bubbles in soft drinks are due to carbon dioxide, which dissolves in water to form carbonic acid.
Every liquid you see will probably have either acidic or basic properties. One exception might be distilled water. Distilled water is just water. The acidic and basic ions in distilled water are in equal amounts and cancel each other out. Most water you drink has an excess of acidic or basic ions in it.
Acids may occur naturally while some are synthetic. They are all characterised by their distinctive sour taste, such as in lemon juice or vinegar and some insect stings, although in many cases it is not recommended to taste them. Not all acids are dangerous but they can be corrosive and dissolve ('eat-away') a number of solid material such as metal, glass, rocks, clothing or burn skin.
The photo below shows some of the many natural acids found in food and drink. For example, The bubbles in soft drinks are due to carbon dioxide, which dissolves in water to form carbonic acid.
Unlike acids, bases taste bitter and feel soapy on your skin. Like acids, some bases can be corrosive, particularly for living tissues such as skin. Bases are found in a variety of home products such as oven cleaner, floor cleaner, drain cleaners, some toothpastes and soda bicarbonate (bicarb soda). Baking powder also contain a base, sodium carbonate. Another word for base is alkali.
2. indicators and pH scale
Any solutions using water as a solvent can be acidic, basic or neutral. A quick way to tell if a solution is acidic or basic is to use an acid-base indicator and a pH scale.
An indicator is a substance which indicates the presence of something. In this case, acid-base indicators change colour to indicate whether a solution is acidic, basic or neutral.
The pH scale is a scale ranging from 0 to 14 which tells us how acidic or basic a solution is. Acidic solutions have a pH less than 7 and bases have a pH greater than 7. A pH of 7 indicates the solution is neutral. There is a definite relationship between pH and amount of H+ ions: the lower the pH of a solution, the more acidic it is, and therefore the more H+ ions it contains.
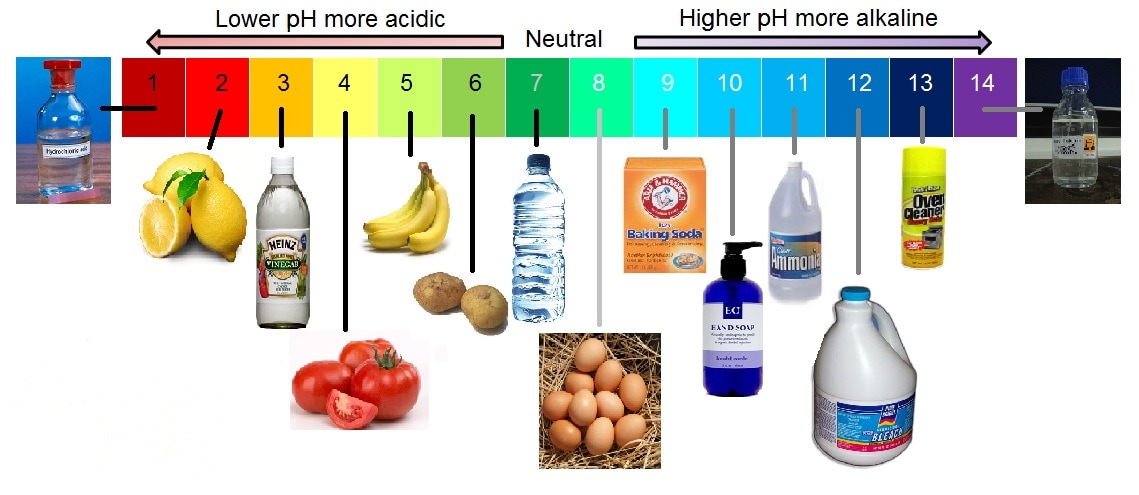
An acid-base indicator is used to estimate the pH of a solution by matching the colour to a pH scale. This can be done using indicators that have colour scales that have been created for that particular indicator. The picture below shows the pH of common substances and whether it is more acidic or alkaline (another word for basic).
An indicator is a substance which indicates the presence of something. In this case, acid-base indicators change colour to indicate whether a solution is acidic, basic or neutral.
The pH scale is a scale ranging from 0 to 14 which tells us how acidic or basic a solution is. Acidic solutions have a pH less than 7 and bases have a pH greater than 7. A pH of 7 indicates the solution is neutral. There is a definite relationship between pH and amount of H+ ions: the lower the pH of a solution, the more acidic it is, and therefore the more H+ ions it contains.
An acid-base indicator is used to estimate the pH of a solution by matching the colour to a pH scale. This can be done using indicators that have colour scales that have been created for that particular indicator. The picture below shows the pH of common substances and whether it is more acidic or alkaline (another word for basic).
Indicators may occur naturally (in plants, e.g. flowers or cabbage) or be synthetic (man-made).
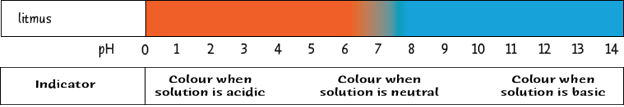
Most indicators only show two colour changes. An example is Litmus. It is an indicator made from lichen (a plant and a fungi that grow together on trees and rocks). Litmus turns red in an acid and blue in a base.
Most indicators only show two colour changes. An example is Litmus. It is an indicator made from lichen (a plant and a fungi that grow together on trees and rocks). Litmus turns red in an acid and blue in a base.
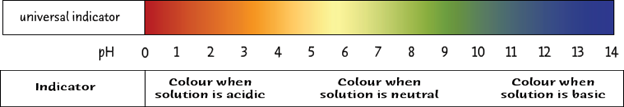
Universal indicator, however, shows many different colours depending on the pH. his is because it is made of a mixture of indicators.
3. safety precautions about acids & bases
- Acids and bases are corrosive, even if classified as weak.
- Eye and hand protection should be worn when using acids and bases. Use rubber gloves when using concentrated acids and bases.
- When diluting an acid or a base, ALWAYS ADD THE ACID OR THE BASE TO WATER and stir while adding.
- Diluting acids and bases releases a large amount of heat.
- Any spill or splashes on the skin must be washed with plenty of running water.
4. chemistry of acids and bases
What are acids?
Acids are compounds that have a hydrogen ion connected to some other ion. Because the hydrogen ion is a positively charged ion, the opposite ion is a negative charged.
One of the most well-known acids out there is hydrochloric acid. Its chemical makeup is HCl, where a hydrogen atom and a chlorine atom are linked by a covalent bond (sharing of a pair of electrons). However, what Arrhenius was suggesting was that, when putting the acid into water, it would split into two charged species. It would look like this:
Acids are compounds that have a hydrogen ion connected to some other ion. Because the hydrogen ion is a positively charged ion, the opposite ion is a negative charged.
One of the most well-known acids out there is hydrochloric acid. Its chemical makeup is HCl, where a hydrogen atom and a chlorine atom are linked by a covalent bond (sharing of a pair of electrons). However, what Arrhenius was suggesting was that, when putting the acid into water, it would split into two charged species. It would look like this:
In other words, Arrhenius said that mixing HCl with water makes a hydrogen ion and a chloride ion. He then went on to answer question which is…
What are bases?
A base is a compound that has a hydroxide ion connected to some other ion. Because the hydroxide ion is negatively charged, the opposite ion is a positively charged ion. One of the most well known bases is sodium hydroxide. Its chemical makeup is NaOH, but unlike HCl, it is an ionic solid where a sodium ion Na+ attracts and is attracted by the negative hydroxide ion OH-. The reason the hydroxide is negative is because, while the hydrogen has a +1 charge, the oxygen has a -2 charge. Combine those together and you get a -1 charge for the ion. Arrhenius suggested that, when putting a base in water, it would split the two apart. It would look like this:
What are bases?
A base is a compound that has a hydroxide ion connected to some other ion. Because the hydroxide ion is negatively charged, the opposite ion is a positively charged ion. One of the most well known bases is sodium hydroxide. Its chemical makeup is NaOH, but unlike HCl, it is an ionic solid where a sodium ion Na+ attracts and is attracted by the negative hydroxide ion OH-. The reason the hydroxide is negative is because, while the hydrogen has a +1 charge, the oxygen has a -2 charge. Combine those together and you get a -1 charge for the ion. Arrhenius suggested that, when putting a base in water, it would split the two apart. It would look like this:
In other words, mixing water with NaOH results in a sodium positively charged ion and a hydroxide negatively charged ion.
Note: the small symbols next to chemical species in the two equations above indicate its state. (s) indicates it is in the solid state and (aq) tells us it is in solution in water. Other symbols are (l) and (g) for liquid and gaseous states respectively.
Note: the small symbols next to chemical species in the two equations above indicate its state. (s) indicates it is in the solid state and (aq) tells us it is in solution in water. Other symbols are (l) and (g) for liquid and gaseous states respectively.
5. neutralising acids
Neutralization is a reaction in which an acid and a base cancel each other out to form a salt and water. During neutralisation, the reactive H+ ion is be turned into the less reactive water molecule H2O by reacting with the base OH-.
The salt is made up of the ions Na+ and Cl- that did not take part in the neutralization reaction.
Acids can also be neutralised using carbonates, which is a type of base:
Acids can also be neutralised using carbonates, which is a type of base:
A practical application of neutralisation is if you have been stung by an insect. The venom from the sting could be an acid or a base (depending on the insect). Whichever it is, you can neutralize it by using the opposite (e.g. vinegar would neutralize a basic sting while bicarbonate soda will neutralize an acidic sting).
6. Concentration and strength of acids and bases
What makes an acid or a base more or less corrosive depends on two factors: how much of it there is (its concentration) and its 'strength'. Although these terms are often confused, they represent two different properties of acids and bases. Before taking a detailed look at these two important ways to characterise acids and bases, watch the video below.
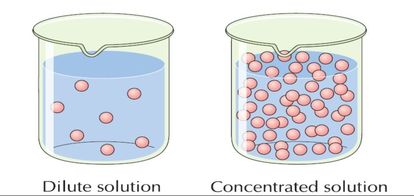
As explained in the video, concentration has to do with the amount of something dissolved in water. You can eat fruit that contain acids because the acid is very dilute, meaning that the fruit does not contain too many acidic ions in comparison to the amount of water the acid is dissolved in. A dilute acid is one that contains a large amount of water and a small amount of acid, typically up to 5% of acid in water.
The same is true for a base. Some household cleaners contain dilute ammonia (a base), which is a can be used to remove grease from floors or to clean windows. Bleach contains dilute sodium hydroxide (base, ca. 0.4%).
The opposite of dilute is concentrated. A concentrated acid is one that contains a percentage of acid in water larger than 5%. The same is true for a base. An example of concentrated acids is the sulfuric acid in a car battery that is highly corrosive and must be handled with great care. Drain cleaners used at home contain concentrated base, sodium hydroxide, that dissolves any organic matter such as hair and grease. They must also be handled with care as they are corrosive to the skin and the eyes.
The picture on the left hand side below illustrated the difference between concentrated and dilute. A dilute solution has a small amount of solute in comparison to the amount of solute in a concentrated solution.
The same is true for a base. Some household cleaners contain dilute ammonia (a base), which is a can be used to remove grease from floors or to clean windows. Bleach contains dilute sodium hydroxide (base, ca. 0.4%).
The opposite of dilute is concentrated. A concentrated acid is one that contains a percentage of acid in water larger than 5%. The same is true for a base. An example of concentrated acids is the sulfuric acid in a car battery that is highly corrosive and must be handled with great care. Drain cleaners used at home contain concentrated base, sodium hydroxide, that dissolves any organic matter such as hair and grease. They must also be handled with care as they are corrosive to the skin and the eyes.
The picture on the left hand side below illustrated the difference between concentrated and dilute. A dilute solution has a small amount of solute in comparison to the amount of solute in a concentrated solution.
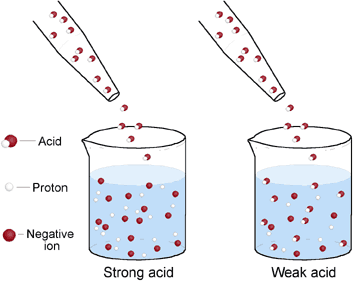
Strength of acids and bases is a more subtle concept that will be explained in detail in Year 11 Chemistry. In simple terms, it has to do with how much of the active form of an acid or base (as ions) becomes available when dissolved in water. For example, if two acidic solutions are made from the same amount of acetic acid (acid making up in vinegar) and hydrochloric acid (use in swimming pools to lower pH), they will have the same concentration. However, the solution containing hydrochloric acid with be stronger than the solution of acetic acid as only 2% of all acetic acid molecules will actually release an acidic ion H+.
In the picture above (right hand side), identical amounts of a strong acid and a weak acid are added to the same amount of water. Although the concentration of each acid is the same in both beakers, the strong acid, which dissociates to a greater extent than the weak acid, produces more H+ ions and negative ions than the weak acid.
The video below is a summary of the important things to remember about acids and bases
In the picture above (right hand side), identical amounts of a strong acid and a weak acid are added to the same amount of water. Although the concentration of each acid is the same in both beakers, the strong acid, which dissociates to a greater extent than the weak acid, produces more H+ ions and negative ions than the weak acid.
The video below is a summary of the important things to remember about acids and bases
7. Antacids
There are many different antacid formulations, over 120 in the United States alone. Most of them contain at least one of these key ingredients: calcium carbonate, sodium carbonate, sodium bicarbonate, magnesium hydroxide and/or aluminium hydroxide. And while all these ingredients work to neutralize your stomach's acid, it is important to know more about them so you can choose the one that's right for you.
Calcium carbonate
Aluminum and magnesium hydroxide
Sodium bicarbonate
Alginate antacids
- Calcium Carbonate is a strong and fast-acting antacid. In fact, it has been used since the first century. And because it's so effective, products like TUMS® still use it today.
- Antacids that contain calcium carbonate may work longer than those containing sodium bicarbonate or magnesium. They also provide calcium, which is lacking in most adult diets. The amount of calcium carbonate usually ranges between 500 and 1,000 milligrams per tablet. It's important to take this calcium carbonate as directed and not exceed the recommendation on the label.
Aluminum and magnesium hydroxide
- Aluminium salts dissolve slowly in the stomach, gradually relieving your heartburn symptoms. But they may cause constipation. Magnesium salts on the other hand, act quickly to neutralize acid but are known to cause diarrhea.
- You could say that two wrongs do make a right. Because the effects of aluminium and magnesium can balance each other out, using them together is often considered an effective treatment for digestive upset.
- However, in recent years there have been questions about the long-term safety of taking aluminium. Because aluminium may deplete the body of phosphorus and calcium (increasing the risk of weak bones) some products are no longer using it.
Sodium bicarbonate
- Sodium bicarbonate is frequently found in products like Alka-Seltzer®, baking soda, or as store brand sodium bicarbonate. It can work quickly to relieve heartburn symptoms. But it's also quickly eliminated from your stomach so relief may not last long.
- Because it reacts with stomach acid and can produce a significant amount of carbon dioxide gas, people often belch or get flatulence when using sodium bicarbonate.
- Sodium bicarbonate has high sodium content and is not appropriate for people who are on salt restricted diets or have congestive heart failure, high blood pressure, or kidney problems. Because it provides such a significant amount of sodium, even people who are not sodium restricted should take this into consideration.
Alginate antacids
- Alginate antacids (like Gaviscon®) don't work the way other antacids do. These antacids contain both calcium carbonate and alginic acid. The alginic acid helps form a barrier that floats on top of the acid in your stomach. This barrier helps prevent stomach acid from moving up into your esophagus.
- The calcium carbonate works to neutralize the stomach acid that pushes through the barrier and into your esophagus. However the amount of calcium carbonate in these products is usually less than in other extra strength ant acids.