Atomic structure
'We think there is color, we think there is sweet, we think there is bitter, but in reality there are atoms and a void.'
|
Key ideas and concepts
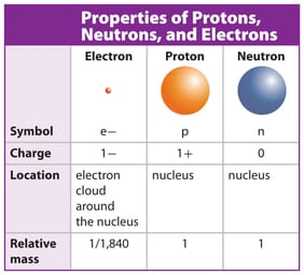
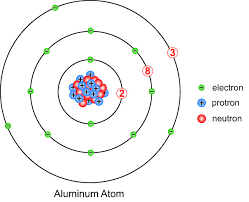
- Atoms are building blocks of matter made of 3 types of subatomic particles: protons, neutrons, and electrons.
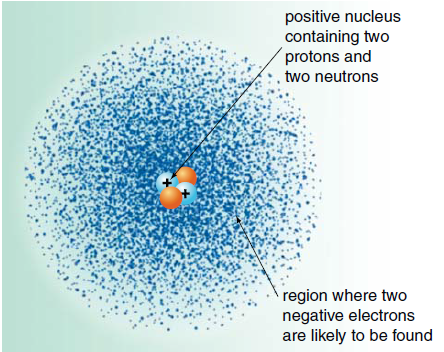
- Protons and neutrons are packed in the centre of atoms, called the nucleus, and concentrate most of the mass of an atom.
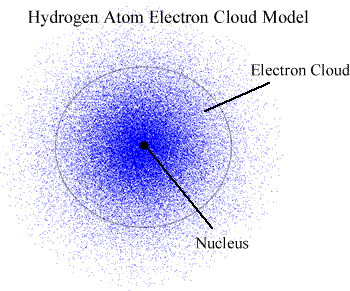
- Negatively charged electrons are attracted to the positively charged nucleus. They move rapidly in the area around it, in well-defined energy levels and can be visualised as fuzzy electron clouds. Their contribution to the mass of an atom is negligible.
- The mass of subatomic particles is expressed in atomic mass unit (u). A proton or a neutron has a mass of about 1 u. The mass of an atom in u is roughly equal to the number of protons+neutrons.
- Protons and neutrons are made of smaller particles called quarks, that were discovered in particle accelerators.
- The LHC is the largest and most powerful particle accelerator and a model of technological achievement. It enabled the discovery of the Higgs Boson in 2013.
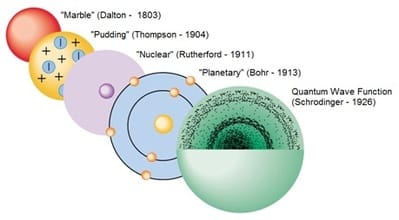
1. Let's start with a bit of history
- 1803: John Dalton introduced a new form of the ancient Greek idea of atoms. He modelled them as hard balls.
- 1897: J.J. Thomson discovered the electron and suggested the 'plum pudding' model of the atom.
- 1911: Ernest Rutherford proposed a new model in which the positive charges in the atom were concentrated in a small central core or nucleus, disproving Thomson's plum pudding model.
- 1914: Niels Bohr modified Rutherford's model by introducing the idea of energy levels, where the electrons move rapidly around the nucleus in fixed orbits, like planets orbiting the sun.
- 1926: Schroedinger developed the wave function that describes the position and state of an electron in an atom in terms of probability, showing that electrons can no longer be thought as orbiting the nucleus at a well defined distance.
- Today, we think of an atom as a positively charged nucleus surrounded negatively charged electrons occupying well-defined energy levels. An energy level can be thought as a region of space where the electrons in that level are most likely to be found, hence the term 'electron cloud' to describe the distribution of electrons in atoms.
For an excellent summary of the history of the atomic model, check this link.
2. anatomy of an atomThree types of sub-atomic particles can be found in an atom:
|
Atomic number and mass number
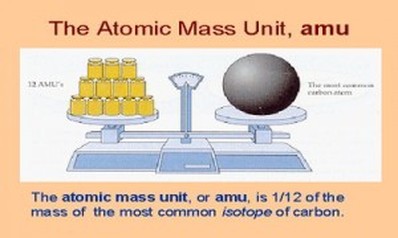
Atomic mass unit
The mass of subatomic particles is extremely small when expressed in kilogram (about one billionth of a billionth of a billionth of kilogram). A more convenient unit is used to express the mass of subatomic particles. It is called atomic mass unit and is represented by the symbol u or amu.
- The number of protons in the nucleus of an atom is called atomic number.
- The number of protons+neutrons is called mass number.
- Protons and neutrons are collectively called nucleons. Therefore, the mass number is sometimes called nucleon number.
Atomic mass unit
The mass of subatomic particles is extremely small when expressed in kilogram (about one billionth of a billionth of a billionth of kilogram). A more convenient unit is used to express the mass of subatomic particles. It is called atomic mass unit and is represented by the symbol u or amu.
- A proton or neutron has a mass of approximately 1 u. An electron is only a fraction of this mass, about 1/1840 u.
- Electrons have so much less mass that they are assumed to have a negligible effect on the mass of an atom.. Therefore the mass of an atom in atomic mass unit is nearly the same as the mass number.
- Example: The mass of a boron atom with 3 protons and 4 neutrons is about 7 u.
Atomic sizes
An atom is roughly a million times smaller than the thickest human hair. That is smaller than a billionth of a meter. (Read the EXtra bits for EXperts section at the bottom of this page for more details on atoms' size.)
The nucleus of an atom is even smaller than an atom: If an atom was enlarged to the size of a football stadium, the nucleus would be the size of a pinhead in the centre of the field and the electrons would be moving rapidly around the stadium. In other words, the diameter of a nucleus is about 10 000 smaller than the diameter of an atom, justifying the statement that an atom, and therefore matter, is mostly empty space (about 99.999999999 %). To understand this number, imagine that you could remove the empty space from the atoms of all people on Earth. The entire human race could then fit in the volume of a sugar cube!
3. Arrangement of electrons
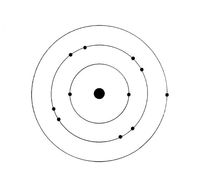
lectrons are arranged around the nucleus in shells or energy levels. Each shell can only contain a certain number of electrons. The maximum number of electrons per shell, in order of increasing shell number (from 1 to 4) is 2, 8, 8, and 18.
The filling order of shells is always from the lowest to the highest shell number. This means that the second shell will not receive electrons until the first shell is completely filled with 2 electrons. Similarly, the third shell will be occupied by electrons only if the second shell is completely filled with 8 electrons.
The electron configuration of an atom or ion is the sequence of numbers that describes the number of electrons found in each successive shell.
The electrons in the outermost shell are called valence electrons. They are of extreme importance as they determine the chemical properties of an element. The outermost shell is therefore called valence shell.
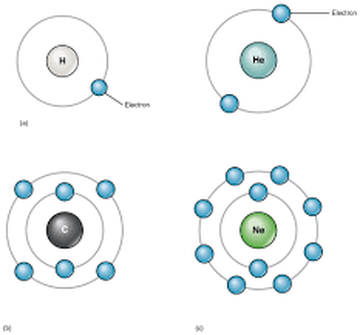
Atoms of the elements hydrogen and helium have respectively one and two electrons in the first shell. Their electron configuration is simply 1 and 2. These elements also occupy the first row of the Periodic Table.
Atoms of the next 8 elements, found in the second row of the Periodic Table, have 2 electrons in the first shell and between 1 and 8 electrons in the second shell, depending on the element. For example, the electron configuration of nitrogen atoms (7 electrons) is 2,5.
Atoms of elements in the third row of the periodic period all have 3 shells of electrons. For example, the electron configuration of magnesium atoms (12 electrons) is 2,8,2. That of argon is 2,8,8.
Watch the video below to consolidate your understanding of how to determine electron configurations.
The filling order of shells is always from the lowest to the highest shell number. This means that the second shell will not receive electrons until the first shell is completely filled with 2 electrons. Similarly, the third shell will be occupied by electrons only if the second shell is completely filled with 8 electrons.
The electron configuration of an atom or ion is the sequence of numbers that describes the number of electrons found in each successive shell.
The electrons in the outermost shell are called valence electrons. They are of extreme importance as they determine the chemical properties of an element. The outermost shell is therefore called valence shell.
Atoms of the elements hydrogen and helium have respectively one and two electrons in the first shell. Their electron configuration is simply 1 and 2. These elements also occupy the first row of the Periodic Table.
Atoms of the next 8 elements, found in the second row of the Periodic Table, have 2 electrons in the first shell and between 1 and 8 electrons in the second shell, depending on the element. For example, the electron configuration of nitrogen atoms (7 electrons) is 2,5.
Atoms of elements in the third row of the periodic period all have 3 shells of electrons. For example, the electron configuration of magnesium atoms (12 electrons) is 2,8,2. That of argon is 2,8,8.
Watch the video below to consolidate your understanding of how to determine electron configurations.
ACTIVITY: for each diagram represented below, write the corresponding electron configuration.
ACTIVITY: for each diagram represented below, write the corresponding electron configuration.
4. Important experiments
Most of what we know about atoms was discovered about 100 years ago, through a number of historical experiments.
The negatively charged electron was the first particle discovered. Later on, evidence that atoms were made of a small and heavy nucleus was produced. It was followed by the discovery of the positively charged proton, one of the two particles making up nucleus. The other nucleus' particle, the neutron, was discovered relatively late due to its lack of electrical charge.
The videoclip below provides an excellent overview on how electrons, protons and neutrons were discovered and by whom. Find out more details on each experiment here (link to the year 10 chemistry weebly).
The negatively charged electron was the first particle discovered. Later on, evidence that atoms were made of a small and heavy nucleus was produced. It was followed by the discovery of the positively charged proton, one of the two particles making up nucleus. The other nucleus' particle, the neutron, was discovered relatively late due to its lack of electrical charge.
The videoclip below provides an excellent overview on how electrons, protons and neutrons were discovered and by whom. Find out more details on each experiment here (link to the year 10 chemistry weebly).
More recently, through the use of particle accelerators, it was discovered that protons and neutrons are themselves made of smaller particles, knwon as quarks. Although there are 6 different types of quarks, protons and neutrons are made up of two types of quarks held together by force particles called gluons. The most powerful and world's largest particle accelerator is the Large Hardon Collider (LHC), a 27 km long circular tunnel located about 100 m underground that accelerates particles to a speed close to the speed of light. The LHC led to the discovery of the Higgs boson in 2012, sometimes referred to as the God's particle.
Most facts associated with the LHC represent scientific and technological achievements in themselves and deal with vertiginous numbers. Click here if you would like to read, hear and learn more about sub-atomic particles, particle accelerators and the LHC (Year 9 student friendly website), or here if you would like to download the corresponding pdf brochure.
Did you know?
Most facts associated with the LHC represent scientific and technological achievements in themselves and deal with vertiginous numbers. Click here if you would like to read, hear and learn more about sub-atomic particles, particle accelerators and the LHC (Year 9 student friendly website), or here if you would like to download the corresponding pdf brochure.
Did you know?
- LHC stands for Large Hadron Collider. Large due to its size (approximately 27 km in circumference), Hadron because it accelerates protons or ions, which are hadrons, and Collider because these particles form two beams travelling in opposite directions, which collide at four points where the two rings of the machine intersect.
- In a standard LHC experiment, there are 600 million collisions every second, generating temperatures more than 100 000 times hotter than the core of the sun.
- The 27-km circumference of the LHC calls for some 7600 km of cable, corresponding to about 270 000 km of strand.
5. it's your turn: Build your own atoms
If you would like to test and enhance your understanding about atoms, read the instructions of the activity sheet below and use the simulation to answer the questions.
EXtra bits for EXperts: Let's talk about size
The size of an atom is most often associated to its radius, usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding cloud of electrons. Because of the boundary-less nature of the electron cloud (similar to an actual fuzzy cloud in the sky), this value cannot be accurately determined and this has led to the fact that there are various non-equivalent definitions of atomic radius. One definition of atomic radius is the region of space where the probability to find the electrons is 90 %.
An analogy could be the earth's atmosphere. As the altitude increases, the density (mass per litre) of the atmosphere decreases and there is no clear boundary where you could say that the atmosphere stops. Similar to the atomic radius, there is no clear agreement on the how thick the atmosphere is. Depending on the source you use, you will find that the thickness of the atmosphere varies between 100 and 480 kilometres, although most of it is it within 16 kilometres.