the periodic table
Key ideas and concepts
- The atomic number of an element is the number of protons. All atoms of the same element have the same atomic number.
- The average mass of every isotope making up an element represents the average atomic mass of the element (or simply atomic mass).
- The name of an element is defined by its atomic number (the number of protons) and is represented by a one- or two-letter symbol.
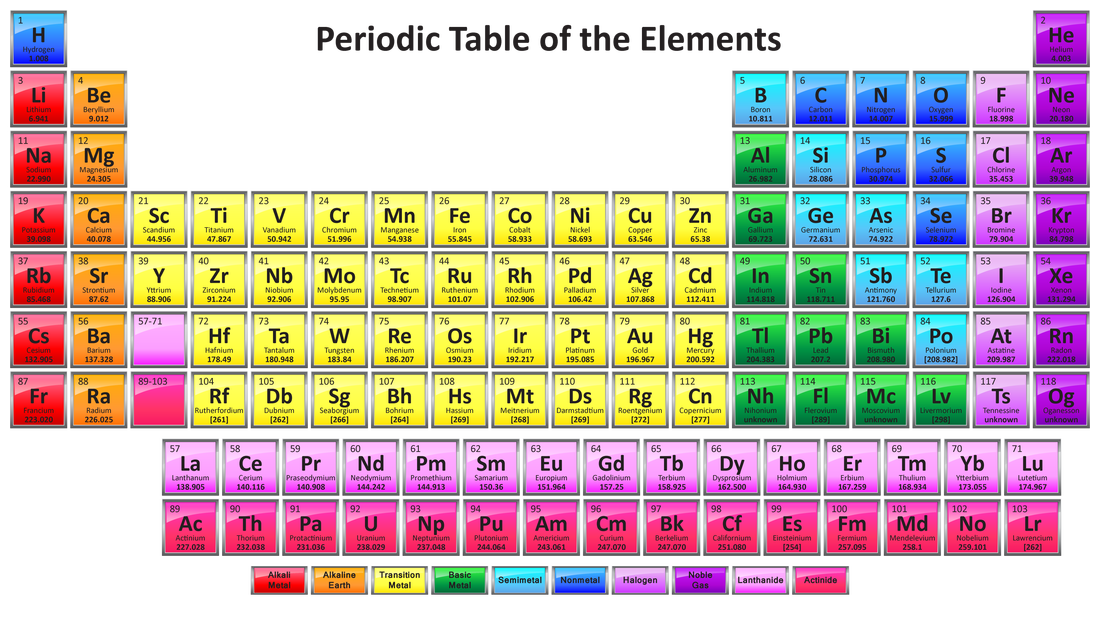
- The Periodic Table is a useful way to arrange the elements in order of increasing atomic number.
- The horizontal rows are called periods; the vertical rows are knwon as groups.
- The elements of the same group have the same number of electrons in their outer shell and have similar chemical properties.
1. arranging the elements
|
Imagine searching through piles of unsorted CDs looking for the latest album of your favourite artist. Such a task could take days. Fortunately music shops arrange their CDs in separate sections. They then divide each of these groups into smaller groups according to the type of music, e.g. rock, popular, jazz and classical. Artists within each group are then arranged alphabetically.
Scientists have a similar problem to the music store owner. There are over 100 different elements, each with different physical and chemical properties. |
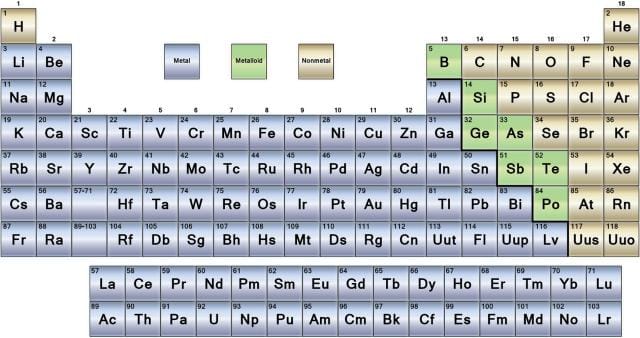
- The periodic table is simply a list of the chemical elements arranged in order of increasing atomic number. Reading like a book from left to right and top to bottom, the atomic number increases in single units from 1 to 118.
- The arrangement is such that similar elements are found next to one another in repeating cycles or periods. From this pattern comes the term 'The Periodic Table'. A number of trends in chemical and physical properties of the elements can be recognised and will be discussed in the next section.
- More than just a list of elements, the Periodic Table is one of the most useful datatables in all of science, providing information on the chemical and physical properties of elements, depending on their position in the table.
- The most common layout of the Periodic Table is shown below, although alternative layouts have been proposed. Click here to obtain more information on different presentations of the Periodic Table.
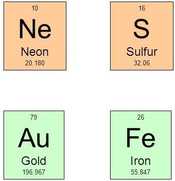
- The symbol of the element always consists of one or two letters, for example Ne for neon or S for sulfur. It does not always tell what the element is, for example Au for gold or Fe for iron.
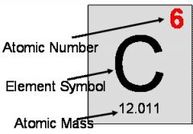
- The smallest of the two numbers (always a whole number) in the box is called the atomic number. It is equal to the number of protons. for example, the atomic number of neon is 10 because all neon atoms have 10 protons in their nucleus.
- The largest number is called the average atomic mass, or atomic mass of the element, expressed in amu (atomic mass unit, symbol u). It is most often a decimal as it is the average of the atomic mass of each isotope making up the element. For example, the atomic mass of iron is 55.847 u because iron consists of a mixture of stable isotopes with different relative abundance so that their average mass is 55.847 u.
|
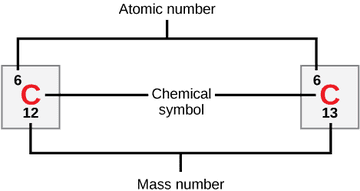
Above is the element carbon as represented in the Periodic Table. The atomic mass is the weighed average of the mass number of its stable isotopes, C-12 and C-13, in atomic mass units. As C-12 is by far the most abundant of the two, the atomic mass of carbon is close to 12.
|
3. Structure of the Periodic Table
The elements of the periodic table can be broken into three major categories, according to their physical and chemical properties: metals, metalloids (or semi-metals) and nonmetals.
The periodic table below shows the three different categories of elements. The metalloid group separates the metals from the nonmetals. Elements to the left are metals and nonmetals are to the right. The exception is the element hydrogen. Hydrogen has properties of a nonmetal at normal temperatures and pressures while it has metallic properties in the solid state.
- Metals are generally shiny, highly conducting solids (except mercury which occurs as a liquid) that form alloys with one another and salt-like ionic compounds with nonmetals (other than the noble gases).
- Nonmetals are solids or gases at atmospheric pressure and ambient temperature, with the exception of bromine which occurs as a liquid. They are insulatorsinsulating gases; nonmetals form compounds with other nonmetals through covalent bonding.
- In between metals and nonmetals are metalloids, which have intermediate or mixed properties.
The periodic table below shows the three different categories of elements. The metalloid group separates the metals from the nonmetals. Elements to the left are metals and nonmetals are to the right. The exception is the element hydrogen. Hydrogen has properties of a nonmetal at normal temperatures and pressures while it has metallic properties in the solid state.
Periods
The horizontal rows, or periods, give the periodic table its name. Each period represents the elements in which the same electron shell is being filled by electrons, until it is full.
Thus, the second period has elements with an incomplete second energy level (except for neon, in which the second level is now full).
Thus, the second period has elements with an incomplete second energy level (except for neon, in which the second level is now full).
Groups (or families)
The vertical columns of elements are known as groups, numbered from 1 to 18.
Some of the groups are also referred to by group names (in times past, they all had names). The most well-known are:
Group number 1: alkali metals
Group number 2: alkaline earth metals
Group number 17: halogens
Group number 18: noble gases
Elements of the same group share similar chemical properties owing to the similarity of electronic configuration in terms of the outer (valence) electron shell. For example, elements of group 1 - the alkali metals - all have one electron in their outer shell. They are all soft metals, which react easily with water.
Some of the groups are also referred to by group names (in times past, they all had names). The most well-known are:
Group number 1: alkali metals
Group number 2: alkaline earth metals
Group number 17: halogens
Group number 18: noble gases
Elements of the same group share similar chemical properties owing to the similarity of electronic configuration in terms of the outer (valence) electron shell. For example, elements of group 1 - the alkali metals - all have one electron in their outer shell. They are all soft metals, which react easily with water.
4. History of the modern periodic table
The Periodic Table is more than just a list of elements. It was the result of investigations and thought by many notable scientists. Its clever structure makes it one of the most useful datatables in all of science, providing information on the chemical and physical properties of elements, depending on their position in the table. Hence its ubiquitous place of honour on laboratory walls.
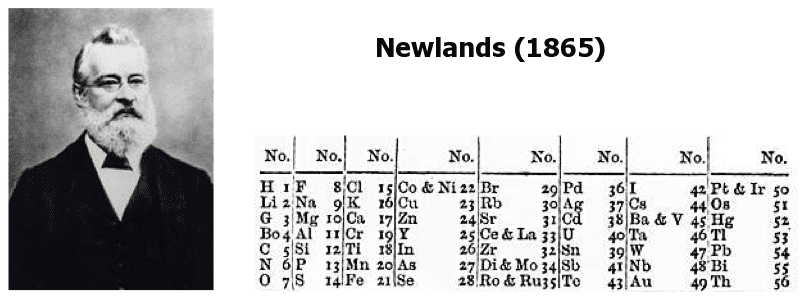
- An English scientist called John Newlands put forward his law of octaves in 1864. He arranged all the elements known at the time into a table in order of relative atomic mass. A reproduction of the table he published in 1865 is represented below. When he did this, he found that each element was similar to the element eight places further on. For example, starting at Li, Be is the second element, B is the third and Na is the eighth element
- Newlands' table showed a repeating or periodic pattern of properties, but it had problems. For example, he put iron in the same group as oxygen and sulphur, which are two non-metals. As a result, his table was not accepted by other scientists.
- In 1869, the Russian chemist Dmitri Mendeleev published a periodic table. Mendeleev also arranged the elements known at the time in order of relative atomic mass, but he did some other things that made his table much more successful. He realised that the physical and chemical properties of elements were related to their atomic mass in a 'periodic' way, and arranged them so that groups of elements with similar properties fell into vertical columns in his table. Below is the world’s first view of Mendeleev’s periodic table published in 1869.
- Mendeleev famously left some empty spots in his early periodic table for yet undiscovered elements (most notably elements that he named eka-boron (Eb), eka-aluminium (Ea), eka-manganese (Em), and eka-silicon (Es), later to be renamed as scandium, gallium, technetium and germanium upon their discovery) and predicted some of their physical and chemical properties. His predictions were later found to be in close agreement with experimental measurements. The table below compares his prediction with experimental measurements for the element gallium, discovered in 1875. More information on the history of the Periodic Table can be found here.