What is matter?
1. classification of matter
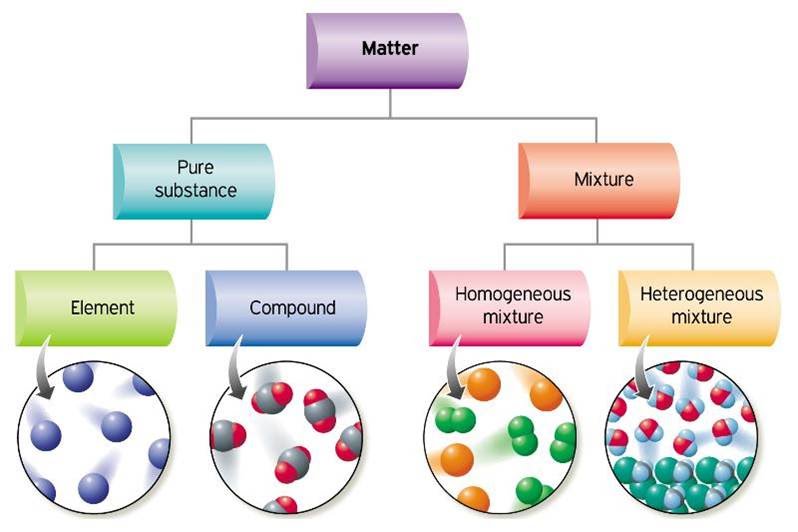
All ordinary matter is made of atoms and can be classified into the categories shown below.
A pure substance can be either an element or a compound.
Two or more pure substances that can be easily separated by physical means form a mixture. There can be two types of mixtures.
Click on the image above to obtain more information on the classification of matter.
- When all the atoms in a substance are alike, the substance is an ELEMENT. For example, the gas oxygen is made of oxygen atoms.
- A COMPOUND is a substance with two or more elements combined in a fixed proportion. For example, chalk is a compound made of carbon, calcium and oxygen atoms. Water is made of hydrogen and oxygen.
Two or more pure substances that can be easily separated by physical means form a mixture. There can be two types of mixtures.
- A mixture of different and easily distinguishable materials, such as concrete, wood, or a soup is a HETEROGENEOUS MIXTURE.
- A HOMOGENEOUS MIXTURE contains two or more gaseous, liquid, or solid substances blended evenly, for example salt water, air or polymers.
Click on the image above to obtain more information on the classification of matter.
2. States of matter
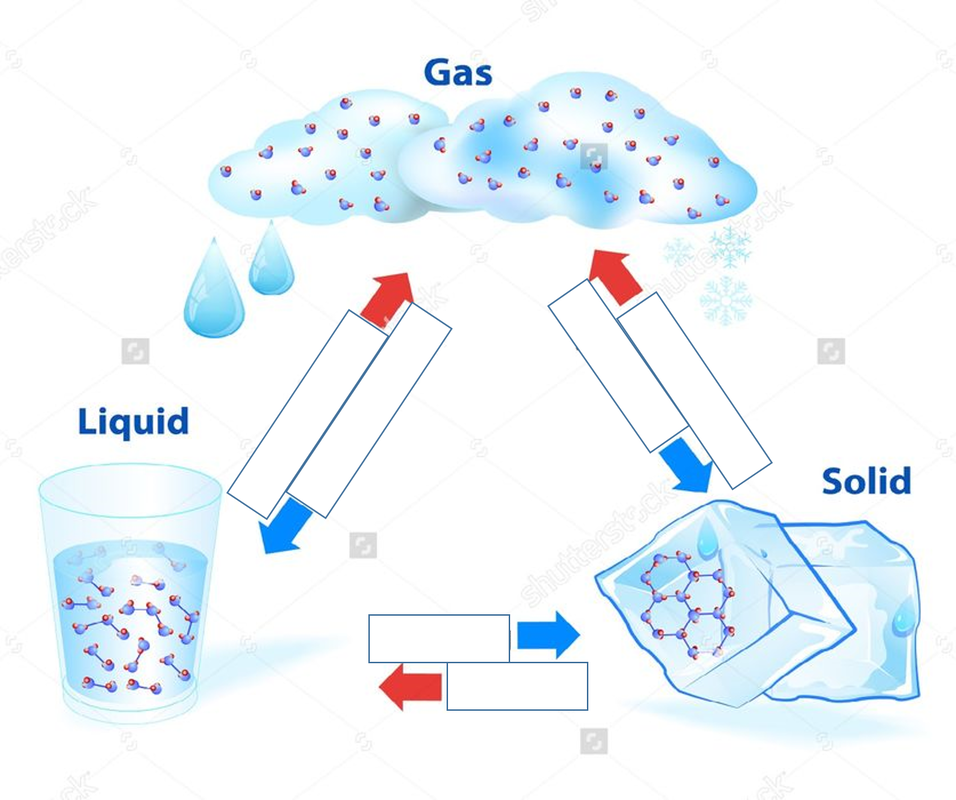
A state of matter is one of the distinct forms that matter takes on. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma.
- Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place.
- Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely.
- Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place.
- Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Plasma is the most common form of visible matter in the universe.
Any transition between two states of matter involves an energy change, that can result in a temperature change. For a solid to melt or a liquid to evaporate, it must receive heat from its surroundings. That's why most metals are solid at room temperature as there is not enough heat to turn them into a liquid. And this is why sweating helps our body to cool down: when the water component of sweat evaporates, it absorbs heat from our skin.
Conversely, as a gas condenses into a liquid or as a liquid freezes into a solid, it releases energy into the environment. Have you ever noticed how hot the back of fridge can be? Some of that heat comes from a gas condensing into a liquid.
These facts can be understood in terms of the Particle Theory. Visit the Science 8 Weebly http://saintschemistry8.weebly.com/states-of-matter.html for a refresher on the Particle Theory.
Conversely, as a gas condenses into a liquid or as a liquid freezes into a solid, it releases energy into the environment. Have you ever noticed how hot the back of fridge can be? Some of that heat comes from a gas condensing into a liquid.
These facts can be understood in terms of the Particle Theory. Visit the Science 8 Weebly http://saintschemistry8.weebly.com/states-of-matter.html for a refresher on the Particle Theory.
Challenge: Can you name the transitions between the solid, liquid and gaseous states of matter? Give it a try and click here to find the answers.
3. Physical and chemical changes
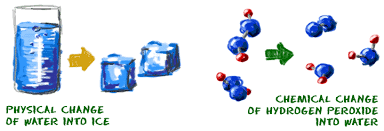
During physical changes, the nature of the particles that make up matter – atoms or molecules – does not change. These particles shift to new positions or change their speed. If they gain energy the particles move more quickly and if they lose energy they move more slowly.
Chemical changes, also known as chemical reactions results in the formation of new substances, called products, from the starting substances, called reactants.
Chemical changes, also known as chemical reactions results in the formation of new substances, called products, from the starting substances, called reactants.
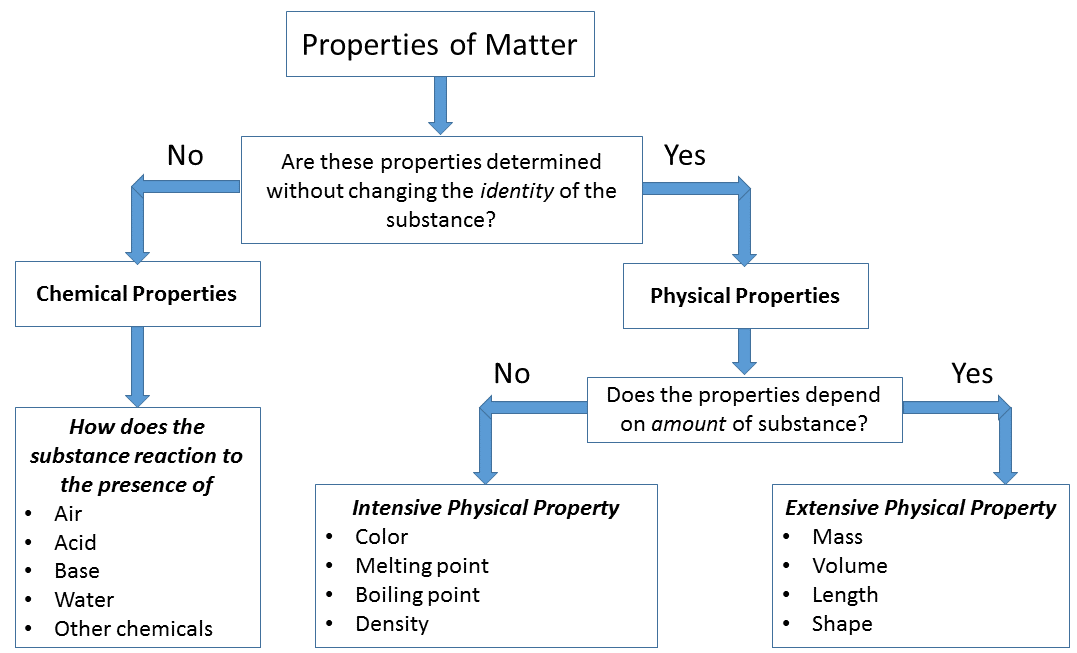
chemical vs physical property
|
Physical properties
They are measured or observed without changing the identity of material. Examples: colour, odour, melting point, boiling point, density |
Chemical properties
They are observed only after chemical change occurs. Examples: acidity or basicity, combustibility |